Registered Address
Chandigarh-160035, India
Work Hours
Monday to Friday: 9AM - 6:30PM


We must accept finite disappointment but never lose infinite hope
– Martin Luther King

Accepting and publishing research work such as original articles, review articles, case series/ reports or letters to an editor in a reputable journal is the most important goal of any medical writer that not only improves career growth but also helps to build one’s confidence. To make sure that the manuscript should not get rejected outright, it is important to create an ideal submission package that includes the already formulated hypotheses, determined methods, accumulated materials, established results, and properly justified conclusions.
In order to expedite the submission process and prevent potential roadblocks to receive a positive response from the journal editor(s), a final submission checklist is required to assist in organizing, chronologizing, and prioritizing each component of the article preparation.
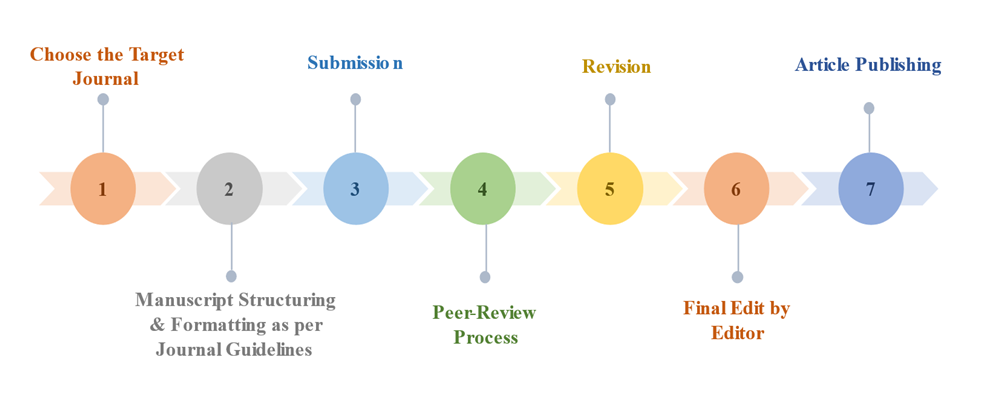
Stepwise Approach in Publishing the Manuscript
CHOOSE THE TARGET JOURNAL
While selecting the journal, one should appropriately go through the journal guidelines. Impact factor, scope, and indexing during journal selection are the important points to be checked. Apart from these, target audience, acceptance and rejection rate, and review and publication timelines should be taken into consideration.
ATTENTION TO JOURNAL REQUIREMENTS
Manuscript structure: The format and structure of the manuscript, as well as its length, should be strictly followed as per the journal’s instructions. An effective abstract, emphasizing the importance of the research problem in the introduction section, describing the materials and methods in detail, discussing the findings and their implications in the results and discussion sections, a justified conclusion, and appropriate references are the key components of any manuscript. The arrangement of these components should be followed exactly as stipulated in the journal standards. This helps to develop coherence and allows readers to better understand the study.
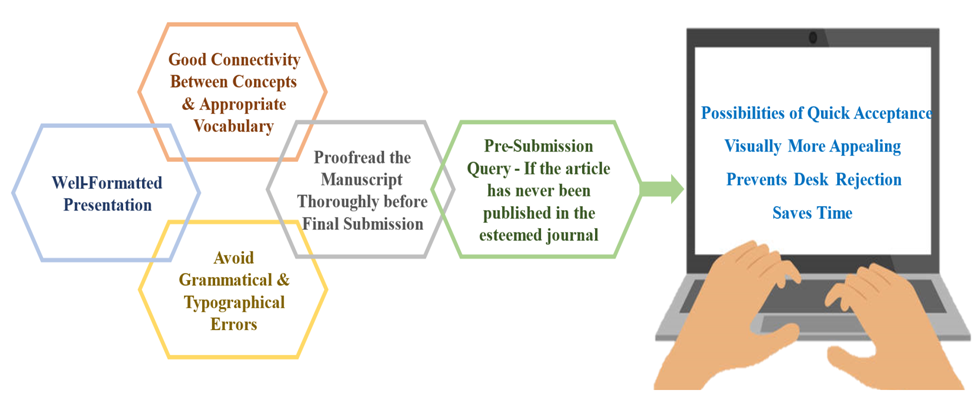
Graphics: Tables and figures should be complete, clear, accurately categorized, neatly produced, and visually appealing. Always take file size, clarity, picture resolution, table titles, figures, graph legends, and captions into account. Material, such as large tables, additional figures, appendices, video, and audio files, should be provided in the separate supplementary files that can help support and improve scientific research.
Facts and Details: Data quality is the most important essence of any manuscript. In addition, other minor things like numerical consistency between text and table, symbols, units of measurement, and congruence between in-text citations and the reference list should also be considered.
WORK TRANSPARENCY
The Cover letter should be prepared in a clear, succinct, and respectful manner to explain why the study should be published. It includes the manuscript’s relevance and impact, full author details, notification of whether the work has already been published elsewhere, and information about any supplemental material. It should be signed by the appropriate authors. The cover letter is for internal review purpose only and should not be published online.
Author Information: Try to include all the essential information, such as the full names of all authors (main and secondary) with designations, contact details of the corresponding author, and a duly signed copyright form. The authorship criteria should be followed as per the ICMJE guidelines.
Attribution and Acknowledgement: Acknowledge those who have contributed to the study. While submitting the work, remember to attribute sources of information, any formal permission from hospitals, labs, or any other source, and informed consent from patients in the form of written, video-graphed, or verbal agreement. Aside from that, in certain publications, the author(s) must provide the names of potential and opposing reviewers who should or should not review their work. During the submission process, their entire contact information, institutional information, and the rationale for their selection/rejection should be included.
Conflict of Interest: In the case of medical research, there might be a potential conflict of interest from any perspective, such as with financing pharmaceutical corporations. Any commercial or funding arrangement that might be viewed as a conflict of interest, should be disclosed. Also, a declaration should be provided if there is no possible conflict of interest.
Compliance: Always ensure that the study follows appropriate ethical standards, such as patient and animal care guidelines, clinical trial registration guidelines, environmental protection guidelines, and an approval number/ID. Most reputable journals reject a paper if sufficient evidence of informed consent is not included.
MANUSCRIPT FORMAT
All journal specifications, such as page margins, page numbering, spacing, in-text citations, references, and required word count and format (Word/LaTeX/PDF) for the abstract and main file, should be carefully followed.
QUICK TIPS
Written by:
Dr. Madhuri Mukhe